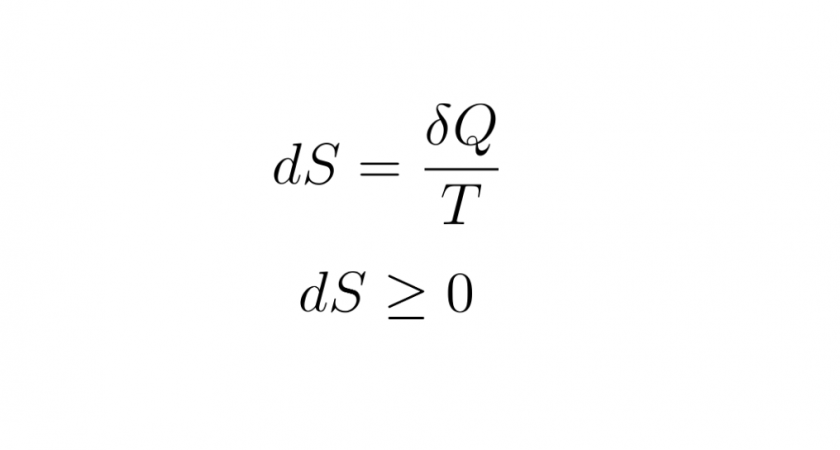The total energy of an isolated system remains constant.
read moreAn equation of state is a mathematical relationship that describes the physical properties of a substance, such as its pressure, volume, temperature, and internal energy, in terms of the state variables that characterize its condition. An equation of state typically takes the…
read moreAdiabatic means heat does not enter or leave the system concerned.
read moreThe zeroth law of thermodynamics provides for the foundation of temperature as an empirical parameter in thermodynamic systems and establishes the transitive relation between the temperatures of multiple bodies in thermal equilibrium.
read moreWork is the product of force and displacement. It is the measure of how much total energy was transferred to the moving object via its accelerating force.
read moreEntropy is the unavailability of a system’s thermal energy for conversion into mechanical work.
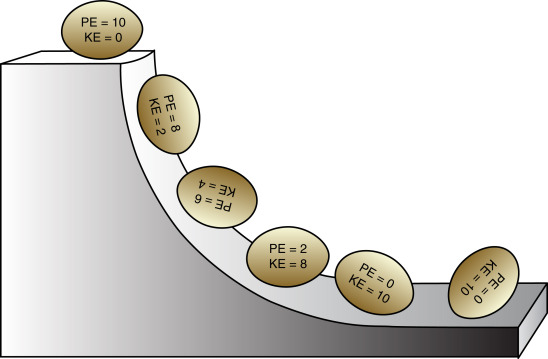
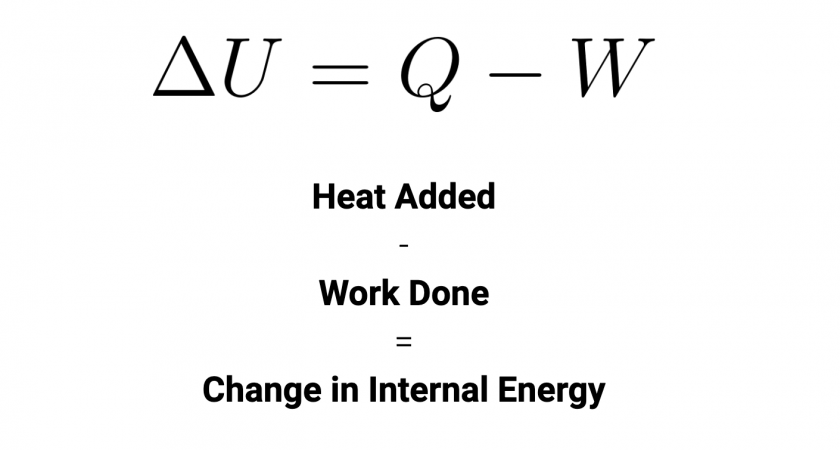
read moreThe Law of Energy Conservation & Transformation The first law of thermodynamics says that energy can neither be created nor be destroyed, it can only be transferred from one form to another.
read moreCosmology is the study of the universe as a whole – its origins, history, and eventual fate; and its matter and energy content as it evolves over time.
read more