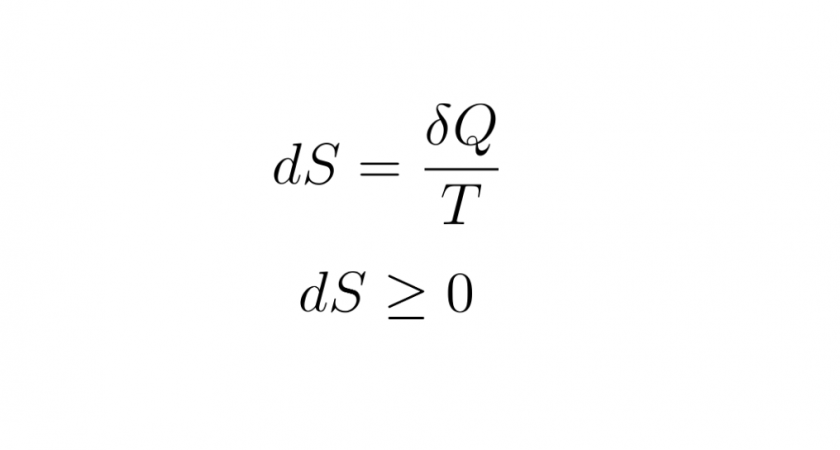Adiabatic means heat does not enter or leave the system concerned.
read moreThe zeroth law of thermodynamics provides for the foundation of temperature as an empirical parameter in thermodynamic systems and establishes the transitive relation between the temperatures of multiple bodies in thermal equilibrium.
read moreEntropy is the unavailability of a system’s thermal energy for conversion into mechanical work.
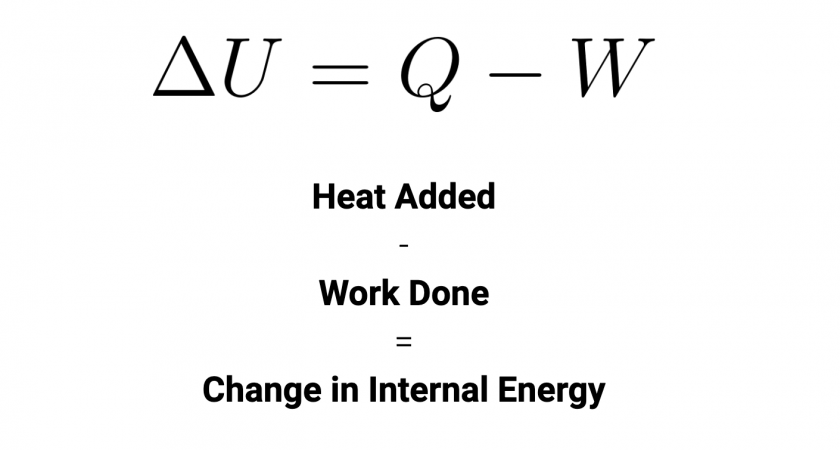
read moreThe Law of Energy Conservation & Transformation The first law of thermodynamics says that energy can neither be created nor be destroyed, it can only be transferred from one form to another.
read more